Accueil du site > Production scientifique > Gas-phase Dissociation Chemistry of Deprotonated RGD
Gas-phase Dissociation Chemistry of Deprotonated RGD
Date de publication: 8 avril 2020
S. Guan ; J. Rabus ; P. Maitre ; B. J. Bythell
J. Am. Soc. Mass Spectrom. 2 89-101 (2020). DOI
Travail réalisé sur le site de l’Université Paris-Saclay.
Abstract
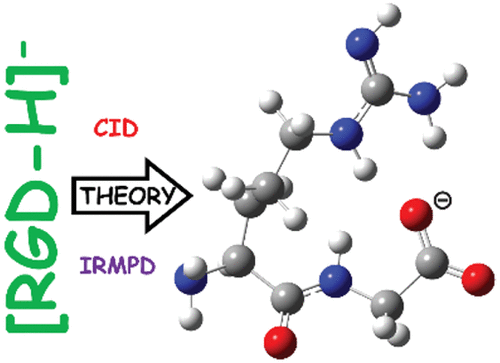
We investigate the structure and dissociation pathways of the deprotonated amphoteric peptide arginylglycylasparic acid, [RGD-H]-. We model the pertinent gas-phase structures and fragmentation chemistry of the precursor anions and predomi-nant sequence-informative bond cleavages (b2+H2O, c2, and z1 peaks) and compare these predictions to our tandem mass spectra and infrared spectroscopy experiments. Formation of the b2+H2O anions requires rate-limiting intramolecular back biting to cleave the second amide bond and generate an anhydride structure through a zwitterion transition structure. Facile cleavage of the newly formed ester bond with concerted expulsion of a cyclic anhydride neutral generates the product struc-ture. IR spectroscopy supports this b2+H2O anion having structures that are essentially identical to C-terminally deprotonat-ed arginylglycine, [RG-H]-. Formation of the c2 anion requires concerted expulsion of CO2 from the aspartyl sidechain car-boxylate with cleavage of the N-Calpha bond to produce a proton-bound dimer of arginylglycinamide and acrylate. Proton transfers enable predominant detection of a c2 anion with the negative charge formally on the central, glycine nitrogen as the proton affinity of this site is predicted to be lower than acrylate by 27 kJ mol-1. Alternate means of cleaving the same N-Calpha bond produce deprotonated cis-1,4-dibut-2-enoic acid z1 anion structures. These processes first involve C-H proton mobilization from the aspartyl sidechain consistent with the literature.








