Accueil du site > Production scientifique > The Histidine Effect. Electron Transfer and Capture Cause Different Dissociations and Rearrangements of Histidine Peptide Cation-Radicals
The Histidine Effect. Electron Transfer and Capture Cause Different Dissociations and Rearrangements of Histidine Peptide Cation-Radicals
Date de publication: 15 juillet 2010
F. Tureek, T. W. Chung, C. L. Moss, J. A. Wyer, A. Ehlerding, A. I. S. Holm, H. Zettergren, S. Brøndsted Nielsen, P. Hvelplund, J. Chamot-Rooke, B. Bythell, B. Paizs
J. Am. Chem. Soc. 132 10728-10740 (2010). DOI
Travail réalisé sur le site de l’Ecole Polytechnique.
Abstract
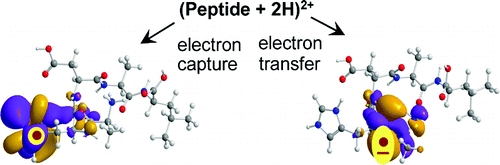
Electron-transfer and -capture dissociations of doubly protonated peptides gave dramatically different product ions for a series of histidine-containing pentapeptides of both non-tryptic (AAHAL, AHAAL, AHADL, AHDAL) and tryptic (AAAHK, AAHAK, AHAAK, HAAAK, AAAHR, AAHAR, AHAAR, HAAAR) type. Electron transfer from gaseous Cs atoms and fluoranthene anions triggered backbone dissociations of all four N−Cα bonds in the peptide ions in addition to loss of H and NH3. Substantial fractions of charge-reduced cation-radicals did not dissociate on an experimental time scale ranging from 10−6 to 10−1 s. Multistage tandem mass spectrometric (MSn) experiments indicated that the non-dissociating cation-radicals had undergone rearrangements. These were explained as being due to proton migrations from N-terminal ammonium and COOH groups to the C-2′ position of the reduced His ring, resulting in substantial radical stabilization. Ab initio calculations revealed that the charge-reduced cation-radicals can exist as low-energy zwitterionic amide π* states which were local energy minima. These states underwent facile exothermic proton migrations to form aminoketyl radical intermediates, whereas direct N−Cα bond cleavage in zwitterions was disfavored. RRKM analysis indicated that backbone N−Cα bond cleavages did not occur competitively from a single charge-reduced precursor. Rather, these bond cleavages proceeded from distinct intermediates which originated from different electronic states accessed by electron transfer. In stark contrast to electron transfer, capture of a free electron by the peptide ions mainly induced radical dissociations of the charge-carrying side chains and loss of a hydrogen atom followed by standard backbone dissociations of even-electron ions. The differences in dissociation are explained by different electronic states being accessed upon electron transfer and capture.








