Accueil du site > Production scientifique > One-step synthesis and reduction of triphenylphosphine carbonyl palladium clusters of variable nuclearities
One-step synthesis and reduction of triphenylphosphine carbonyl palladium clusters of variable nuclearities
Date de publication: 23 avril 2011
C. Willocq, B. Tinant, F. Aubriet, V. Carré, M. Devillers, S. Hermans
Inorg. Chem. Acta 373 233-242 (2011). DOI
Travail réalisé sur le site de l’Université de Lorraine.
Abstract
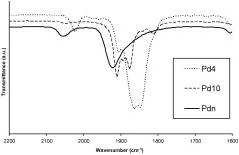
Heteroleptic triphenylphosphine carbonyl palladium clusters of different nuclearities were prepared under mild conditions by only varying the amount of ligand (PPh(3)) used in the synthesis : three different clusters were successfully isolated after CO bubbling in a solution of [Pd(2)(dba)(3)] (dba = dibenzylideneacetone) with 3, 1 or 0.5 equiv of PPh(3), which led, respectively, to [Pd(4)(CO)(5)(PPh(3))(4)] (1), [Pd(10)(CO)(12)(PPh(3))(6)] (2) and [Pd(n)(CO)(x)(PPh(3))(y)] (3) (n approximate to 24). The molecular structures of compounds 1 and 2 were determined by X-ray crystallography. The metal cores in these compounds were shown to consist in a butterfly for 1 and a bridged octahedron for 2. Compound 3 was shown to be at the boundary between molecular clusters and colloidal particles with tentative formulation arising from characterization data. These three clusters and the known [Pd(10)(CO)(12)(PBu(3))(6)] and [Pd(12)(CO)(15)(PBu(3))(7)] were submitted to NaBH(4) reduction. The Pd(4) cluster 1 did not react. The colloidal Pd(n) species led to no isolable product. By contrast, the two Pd(10) and the Pd(12) clusters led to reduction products, isolated as NEt(4)(+) salts. In the case of the reduced Pd(12) cluster, its structure was resolved by X-ray crystallography : the metal core consists of a face-capped octahedron. The reduced species reacted readily with Au(PPh(3))(+), confirming their anionic nature.








